Marco Cavaleri, head of health threats and vaccine strategy at the Amsterdam-based agency, told Rome’s Il Messaggero newspaper on Tuesday that the European Union’s medicines regulator is preparing to make a more definitive statement on the topic this week.
Asked about Mr Cavaleri’s comments, the EMA press office said its evaluation “has not yet reached a conclusion and the review is currently ongoing.”
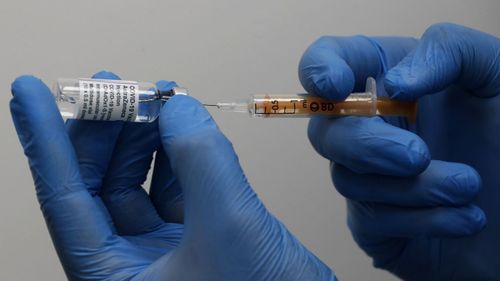
It said it planned a press conference as soon as the review is finalised in coming days.
Based on the evidence so far, Mr Cavaleri said there’s a clear association between the AstraZeneca vaccine and the dozens of rare blood clots that have been reported worldwide amid the tens of millions of AstraZeneca shots that have been given out.
“It is becoming more and more difficult to affirm that there isn’t a cause-and-effect relationship between AstraZeneca vaccines and the very rare cases of blood clots associated with a low level of platelets,” Mr Cavaleri was quoted as saying.
AstraZeneca did not immediately respond to a request for comment.
Late in the day, however, the pharmaceutical company and Oxford University, which developed the vaccine, announced they were pausing the trial of their jabs in children while British regulators investigate the potential blood clot link in adults.
“Whilst there are no safety concerns in the paediatric clinical trial, we await additional information” from the British regulator, an Oxford spokesperson said in a statement.
In Geneva, the World Health Organisation said its experts were also evaluating a possible link between the AstraZeneca vaccine and rare blood clots — and that it might have a “fresh, conclusive assessment” before Thursday.
In March, more than a dozen countries, including Germany, suspended using AstraZeneca over the blood clot issue.

Most EU nations restarted on March 19 — some with age restrictions — after the EMA said the benefits of the vaccine outweighed the risks of not inoculating people against COVID-19.
At the time, the EMA recommended the vaccine’s leaflet be updated with information about the rare clots.
Any further doubts about the AstraZeneca vaccine would be a setback for the shot, which is critical to Europe’s immunisation campaign and a linchpin in the global strategy to get vaccines to poorer countries.
The AstraZeneca vaccine is cheaper and easier to use than rival vaccines from Pfizer and Moderna and has been endorsed for use in over 50 countries, including by the 27-nation EU and the WHO.
United States authorities are still evaluating the vaccine.
Mr Cavaleri said while EMA was prepared to declare a link, further study was still needed to understand why and how the phenomenon occurs.
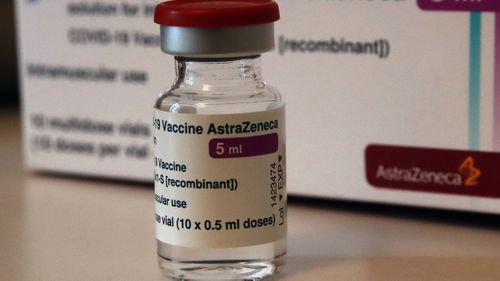
He said the rare blood clots, including some in the brain, coupled with a low level of blood platelets that may make people at risk of serious bleeding, “seem to be the key event to study further.”
Mr Cavaleri promised more details soon, adding, “In the coming hours, we will say that the link is there, how this happens we still haven’t figured out.”
Mr Cavaleri said the biological mechanism for how the vaccine might be causing the rare clots was still unknown and if it was linked to how the shot is made, other vaccines with similar technologies might also need to be evaluated.
He stressed the risk-benefit analysis remained positive for the AstraZeneca jab, even for young women who appear to be more affected by the clots.
“Let’s not forget that young women also end up in intensive care with COVID. So we need to do very meticulous work to understand if the risk-benefit analysis remains for all ages,” he was quoted as saying.
He ruled out a preventive therapy to address the rare blood clots, saying there is still too much unknown about the phenomenon.
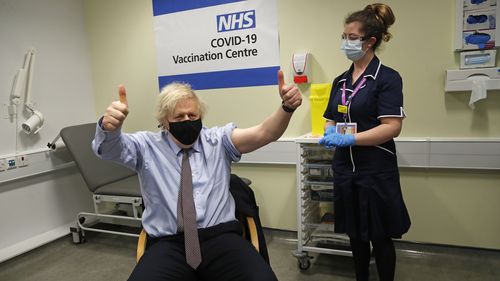
Even after the March 19 restart, the Dutch and German governments suspended the jabs for people under 60 and some Europeans have been shying away from getting a shot.
Romania’s national vaccination committee’s chief, Valeriu Gheorghita, said on Tuesday that since March, 207,000 people in Romania had cancelled their AstraZeneca vaccine appointments and another 92,000 simply didn’t show up.
“It is a high percentage, a third of people scheduled who did not show up,” Mr Gheorghita told reporters.
British Prime Minister Boris Johnson declined to be drawn directly into the latest warnings about the vaccine but urged people to look at the advice from Britain’s independent Medicines and Healthcare Regulatory Agency.
“Their advice to people is to keep going out there, get your jab, get your second jab,” he said during a visit Tuesday to an AstraZeneca facility in Macclesfield, in northwest England.
Last week, Britain’s MHRA said seven people had died in the UK due to blood clots after getting the AstraZeneca jab.
It said it wasn’t clear if the shots are causing the clot and that it was undertaking a “rigorous review” into the reports.
The agency said it had identified 30 blood clot cases out of 18.1 million AstraZeneca jabs given by March 24.
Adam Finn, a professor of paediatrics at the University of Bristol, said the latest surge of COVID-19 cases that is filling up hospitals across Europe should prompt people to get vaccinated as soon as possible.
“If you are currently being offered a dose of Oxford-AstraZeneca vaccine, your chances of remaining alive and well will go up if you take the vaccine and will go down if you don’t,” he said.
This content first appear on 9news

Beneath are three different teres main and minor workout routines that promote balanced energy growth and general shoulder well being.
The 4 finest alternate options to compound shoulder workout routines are listed under.
Contemplate the next Arnold press variations to stop
stagnation in your shoulder workouts. Don’t forget you can use machines, resistance bands, and body weight workout routines,
too.
By combining targeted workout routines, a well-balanced diet, and
adequate recovery, you will be nicely on your way to constructing impressive
shoulder power and muscle bulk. Bear In Mind, consistency and persistence are key, and all the time listen to your
physique to avoid overtraining and accidents. You’ll
discover dozens of full applications to maintain your shoulders growing in the BodyFit by Bodybuilding.com Muscle-Building Exercise Plans.
Once you grasp the routines here, use BodyFit to take your complete upper
body to the subsequent stage. As the ultimate pec train of this chest and shoulder
exercise, you’re going all-out, doing as many reps as you
probably can to failure.
Sometimes, the standing shoulder shrug variations involve heavy training weights, that are lowered because the torso angle changes.
Barre exercises have gained reputation for his or her unique blend of ballet-inspired actions, Pilates strategies, and power training.
For those in search of additional help, Svetness trainers provide professional coaching and customized
exercise plans designed to match your fitness degree and
goals. Customized steerage helps you stay on track and
progress on the proper tempo.
If you’re looking for an excellent exercise to train your posterior chain, there is no higher possibility than the ITY.
Whereas each can provide your upper physique a exercise,
just one won’t have folks taking a look at you sideways within the gym.
Situated at the front of your shoulder joint, the anterior deltoid is concerned
in each overhead and urgent train you do. Its features embrace
flexion and medial rotation of the shoulder joint. Saini suggests protracting your shoulders by rounding your upper back slightly on the backside of the motion to extend the range of motion and maximize target muscle fiber recruitment.
This indicates that the neutral grip may be the higher possibility.
Also, understand that the four rotator cuffs are important muscles that shouldn’t be neglected.
Additionally, as proven in these two research by Trebs and
Lauver, its involvement in pressing movements
will increase considerably as the extent of incline increases.
Are you excited about your subsequent trip but apprehensive about messing up your exercise
routine?
As a veteran personal coach, I can attest that these high-volume exercises can take a ton of time if you’re not
mindful. When you spend too lengthy within the
fitness center, it’s most likely not from lifting too lengthy however
from taking longer-than-needed rest periods between workout routines.
This is counterproductive as it lowers your training depth and total productiveness.
This high-volume exercise resulted in a big pump, and
there have been moments during the aspect lateral raises,
where I felt my shoulders would burst. In this article, I take you thru my expertise
following Jackson’s shoulder workouts and the way they
impacted my muscle dimension and definition and fatigue levels.
I’ll also share tips on how you can undertake this challenge in your individual routine.
Sitting dumbbell shoulder presses are a wonderful way
to build your shoulders, improve posture and stability,
cut back harm risk, and develop power in overhead actions.
Select workout routines that target completely different muscle
groups throughout the shoulders, similar to
combining a compound train just like the barbell overhead press
with an isolation train like lateral raises.
Begin with lighter weights and concentrate on sustaining proper kind throughout the superset.
Lateral deltoid workout routines target and isolate the lateral
deltoid muscle tissue of the shoulders. The beforehand described lateral raises are a type of
lateral deltoid train but not all lateral deltoid workout routines incorporate lateral raises.
For instance, barbell upright rows and Arnold presses are two forms of
lateral deltoid workout routines which might be distinct from lateral raises.
References:
best non steroid supplement (https://78.159.193.219)
70918248
References:
rich piana steroid cycle instagram; socialwavepro.be,